Meet Dr. Susan Majka and Her Lab’s Mission

Dr. Susan M. Majka is a Professor of Medicine at National Jewish Health and a leading researcher in the field of lung regeneration, microvascular biology, and endothelial progenitor cell (mvEPC) function. Her lab is jointly affiliated with the University of Colorado Health Sciences Center and the Gates Center for Regenerative Medicine, where she investigates how endothelial progenitor cells (mvEPCs) support lung health, or contribute to disease progression.
The Majka Lab investigates how mvEPCs regulate the maintenance of the lung’s smallest blood vessels under normal conditions and how their function is disrupted in chronic conditions, including pulmonary fibrosis, emphysema, and LAM. These diseases are often marked by vasculopathy, characterized by progressive loss of microvessels, tissue stiffening, and inflammation, processes where mvEPC dysfunction plays a central role.
By understanding how these progenitor cells behave in both healthy and diseased environments, Dr. Majka’s lab hopes to pave the way for targeted and cell-based regenerative therapies for patients with chronic lung diseases.
A Major Step Forward: 3D Imaging of the Alveolar-Capillary Network
In a recent publication, Thorndyke et al. (American Journal of Physiology – Lung Cell and Molecular Physiology, 2025) from the Majka Lab developed a novel method for visualizing and quantifying lung microvasculature in three dimensions. This workflow fills a critical gap in lung research, where studying the complex, branching capillary network has historically been limited by imaging constraints.
To overcome this, the team combined several key techniques:
- Retro-orbital injection of fluorescent tomato lectin to label pulmonary endothelial cells
- Precision-cut lung slices (PCLS) at 150 µm thickness using the Compresstome® vibratome
- Confocal imaging followed by advanced 3D reconstruction using Imaris software
This innovative approach enables the detailed measurement of vessel density, volume, and branching, providing new insights into the mechanisms of angiogenesis and vascular loss in fibrotic lungs.
How the Compresstome® Vibratome Enables This Research
A cornerstone of this method is the use of the Compresstome vibratome, which enables the Majka Lab to prepare uniform, high-quality precision-cut lung slices (PCLS) while preserving tissue architecture. These slices are thick enough to capture 3D spatial information but thin enough for high-resolution microscopy and dye penetration.
Compared to traditional sectioning methods, the Compresstome offers key advantages for lung tissue preparation:
- Preserves alveolar and vascular microarchitecture without distortion
- Improves reproducibility across tissue sections and experimental replicates
- Maintains tissue viability, enabling live imaging and downstream assays
This level of precision is essential for quantitative vascular imaging and for capturing subtle changes that occur during disease progression.
A Look at the Data: Visualizing Vascular Remodeling
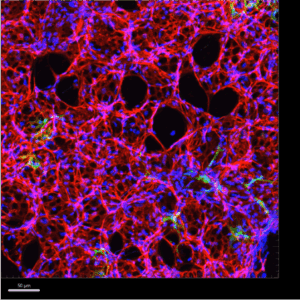
In the Thorndyke et al. study, Dr. Majka’s lab introduced a new workflow for capturing 3D architecture of the lung’s microvasculature in both healthy and fibrotic tissue. Using precision-cut lung slices (PCLS) prepared at 150 µm with the Compresstome vibratome, they performed retro-orbital perfusion with fluorescently labeled tomato lectin to selectively label endothelial cells within the pulmonary vasculature. These slices were then cleared, imaged using confocal microscopy, and reconstructed using Imaris software.
What did this reveal? In control lungs, the vascular network appeared as an intricate web of evenly branched capillaries hugging the alveoli, representing the standard alveolar-capillary interface required for gas exchange. In contrast, lungs from fibrotic mice exhibited pronounced remodeling, characterized by areas of capillary dropout, collapsed alveolar structures, and irregular, tortuous vessel branching patterns.
Importantly, this method enabled quantitative analysis of vessel volume, density, and branching complexity across whole regions of lung tissue—something that is not possible with traditional 2D histological slices. This 3D visualization highlights how microvascular rarefaction contributes to impaired gas exchange and lung stiffening in fibrotic disease.
By using PCLS, the Majka Lab was able to retain enough spatial context to study how fibrosis distorts the vascular niche in situ, offering a level of insight that has direct implications for evaluating disease severity and testing regenerative interventions.
Figure 1. Using the Imaris software, A. 360° rotation and B. orthogonal slicer animations were created from 3D reconstructed z-stacks of 10X images of 150μm thick PCLS for enhanced spatial analysis of the pulmonary microvasculature
This platform now positions the lab to ask critical questions, such as how cell therapies, small molecules, or gene editing tools might restore standard vascular structure in injured lungs.
Looking Ahead: Regeneration, Imaging, and Repair
Dr. Majka’s lab continues to expand this platform to include multiple lung injury models combined with unique transgenic mouse lines, including bleomycin-induced fibrosis and cigarette smoke-induced emphysema. They are also applying the method to study how the lung microenvironment influences progenitor cell differentiation and function, key questions for regenerative medicine.
Future directions include testing candidate cell therapies and investigating how transplanted progenitor cells interact with diseased lung tissue. The Compresstome’s role in producing consistent, quantifiable slices will remain a critical part of these experiments.
Supporting Discovery with Precision Sectioning
We’re honored that the Compresstome vibratome supports Dr. Majka’s groundbreaking work in vascular biology and chronic lung disease. Her lab exemplifies how advanced tools, such as PCLS, can reveal fundamental insights into disease progression and repair.
Want to learn more about PCLS or connect with our featured researchers?
📬 Contact us to get in touch with our team or learn how precision-cut lung slices can support your research.