The Compresstome® vibrating microtome models are unique for many reasons. The major reason is the patented compression technology that is equipped with our slicers only. The second major reason is that, the Compresstome® moves vertically (at a diagonal) to produce a tissue section, opposed to the traditional horizontal method of cutting.
Why is understanding these two aspects important? For one, the patented compression technology is capable through our Compresstome® specimen tubes, which act as the “stage” where your tissue is mounted in order to be cut. After your tissue is adhered to this stage and embedded in agarose (giving the tissue 360˚ stability within the specimen tube, along with accompanying the benefits of the compression advantage), the tissue/specimen tube/stage is then inserted, horizontally, into the Compresstome® buffer tray to be cut, as shown in Figure 1.

As the micrometer advances the tissue forward to be cut, the blade holder moves downward along the cutting path to produce a slice based on the slice thickness reading on the control box. The slice then falls into the buffer tray, and the vibration head unit moves back up to the top of its cutting path, preparing to make an additional cut. This can be seen in the video below.
Step-down Process
As you can see, the Compresstome® is easily able to produce thin, consistent slices. However, you can also tell that there are already many additional sections in the buffer tray. This is due to the step-down process, as the thin slices are not produced at the start of cutting with the Compresstome®. There are two reasons that explain this best:
- The agarose/sample exiting the specimen tube needs to have a heavy-enough weight in order for gravity to pull it outward and downward to be cut. Thus, beginning to section anywhere under 100 µm is far too thin in order for the Compresstome® to produce accurate, reliable sections right from the start.
- The micrometer, which is pushing the plastic plunger of the specimen tube forward (where your tissue sample is on the other end to be cut), is always going to be in a different location at the start of each experiment. There needs to be an equal amount of push (the micrometer) and pull (gravity weighing down on the sample exiting the specimen tube) in order for the Compresstome® to produce a full, accurate slice. It takes some starting slices to reach this equilibrium of the Compresstome® cutting ability, especially when you are producing thinner sections.
Thus, in order to produce full, accurate slices, we highly recommend the step-down cutting process. In order to ensure your tissue sample does not get cut away during the step-down process, we advise drawing down the specimen tube so much that the top of your sample is not right at the specimen tube opening, allowing space to add approximately 2 – 3 mm of agarose above your tissue sample within the specimen tube. This way, only agarose will be cut away during the step-down cutting process, and your tissue will not be cut until you reach your desired slice thickness as the Compresstome® reaches its equilibrium of cutting. An example of leaving extra space on top of the tissue sample for adding extra agarose can be seen in Figure 3 (note that this is a clay brain & epoxy sample of what embedded samples looks like on the specimen tube plunger).

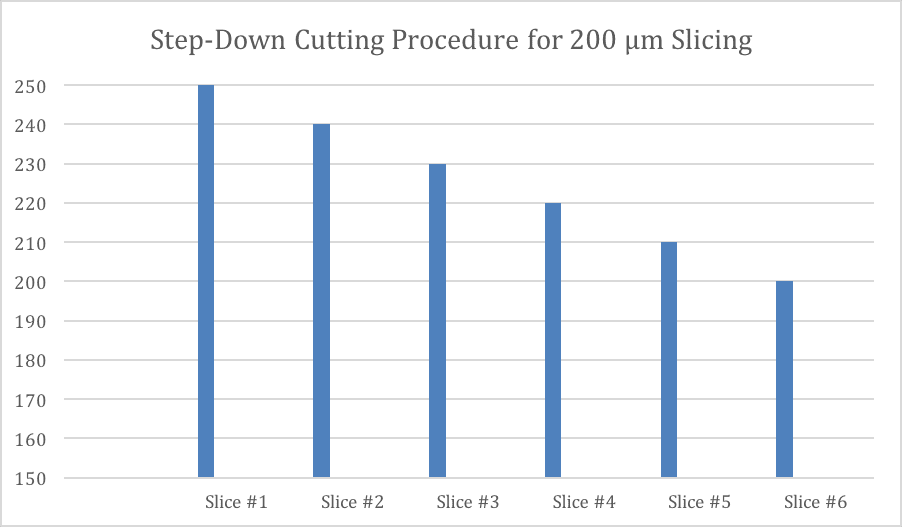
When executing the step-down process, there is a variation between stepping down for a 20 µm slice and a 250 µm slice. Because 250 µm is a heavier weight exiting the specimen tube, there is less of a need to step-down so much, and in so detail. Whereas stepping down for the 20 µm section cutting requires more steps and, more stepping down. This can be seen in Figure 3 and 4 of cutting.
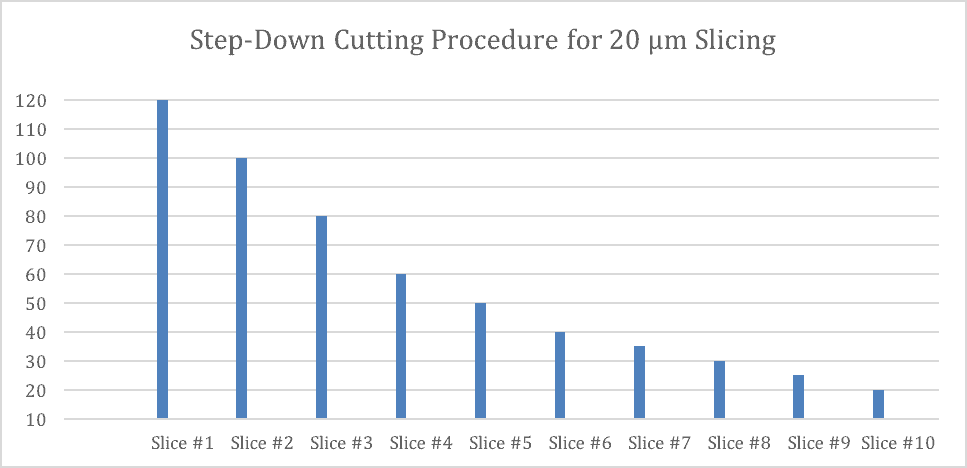
As you can see in Figure 3, cutting thinner sections on the Compresstome® requires a greater stepping down process for obtaining thinner cuts. For thinner sections, especially less than 50 µm, it is best to begin 100 µm greater than your desired slice thickness and step down in units of 20 µm, until you get within 50 µm of your desired slice thickness, to then step-down in 10 µm increments. As you get even closer, within 20 µm, you will want to step-down in 5 µm increments until you reach your desired slice thickness.
As for cutting thicker sections with the Compresstome®, such as 200 µm sections (Figure 4), you do not need to step-down in such greater increments due to the heavier weight exiting the specimen tube already. Seen in the example of Figure 5, you only need to begin cutting 50 µm greater than your desired slice thickness for producing sections that are greater than 100 µm. To produce a 200 µm slice, for example, you only need to begin cutting at 250 µm and step-down in 10 µm increments until you get to your desired slice thickness.
The step-down cutting process is a simple, yet effective way to produce the best tissue slices using the Compresstome®. Executing the step-down cutting process will ensure that your sections are produced at their desired slice thickness every time you cut.
As always, please feel free to reach out to us for more information. We’re always happy to hear from you.