At Precisionary Instruments, we understand that preparing acute brain slices for electrophysiology is a highly technical and delicate process. Many researchers have reached out to us, looking for a clear and structured protocol that walks them through every step. In response, we have developed a detailed, step-by-step experimental guide, one of the first of its kind, that provides a comprehensive overview of acute brain slice preparation, from brain dissection to high-quality patch-clamp recordings.
Electrophysiology is a powerful technique for studying neuronal function, but the quality of data obtained depends heavily on the preparation of brain slices. A well-prepared acute brain slice ensures that neurons remain viable, maintain their physiological properties, and are suitable for patch-clamp recording. This new protocol is designed to help researchers achieve consistently healthy brain slices, reduce variability in experimental outcomes, and streamline the entire process.
Why This Protocol is Important
Patch-clamp electrophysiology is a gold-standard technique for investigating neuronal excitability, synaptic transmission, and ion channel function. While techniques such as calcium imaging and optogenetics have expanded our ability to study brain function, patch-clamp recording remains the most direct and high-resolution method for measuring electrical activity at the single-cell level.
However, achieving high-quality patch-clamp recordings begins with the brain slice preparation process. Poorly cut slices, damage to neuronal membranes, or improper incubation can lead to inconsistent results, making it difficult to obtain reliable electrophysiological data. This guide was developed to address these challenges by providing researchers with a fully detailed and structured protocol that simplifies each stage of the process.
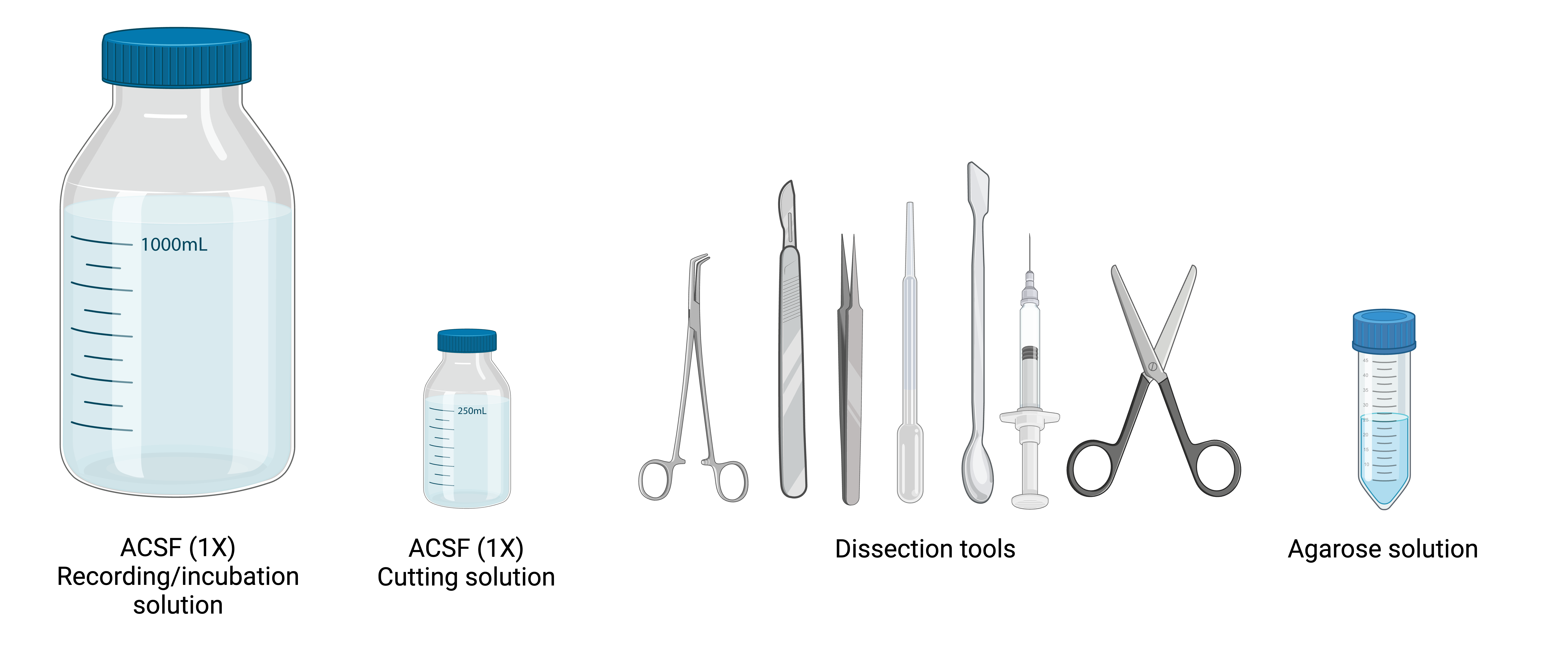
Figure 8. Preparing the Slice Area and Dissection Tools. Ensure all solutions are bubbled with carbogen (95% 02’5% CO2) before slicing, and fill the incubation chamber with cutting solution containing 0.1 mM Ca2+ and 3 mM Mg2+ Set up the slicing area with the necessary tools and equipment as shown. A close-up view highlights essential dissection tools, including fine forceps, razor blades, and a transfer pipette for moving slices. Additionally, a syringe with a bent needle tip, critical for detaching brainstem slices from the cerebellum, is detailed.
What This Guide Covers
This experimental guide is a complete, step-by-step manual that takes researchers through every aspect of acute brain slice preparation and electrophysiology experiments. Key sections include:
- Mouse brain dissection for electrophysiology – A clear, structured approach to removing the brain while preserving tissue integrity.
- Embedding and sectioning with the Compresstome vibratome – A critical guide to obtaining high-quality slices while minimizing tissue damage.
- Optimizing slicing conditions – Including buffer solutions, cooling techniques, and troubleshooting tips for preventing excitotoxicity.
- Patch-clamp setup and electrode positioning – Instructions for achieving successful gigaseals, whole-cell recordings, and single-channel recordings.
- Electrophysiology troubleshooting – Solutions for common challenges such as high access resistance, unstable baseline recordings, and noise reduction strategies.
This protocol is intended for both new and experienced researchers. For those new to electrophysiology, it provides a foundational step-by-step guide, ensuring that critical aspects of the experiment are carried out correctly. For more experienced scientists, it serves as a troubleshooting resource with advanced tips for improving the quality and consistency of recordings.
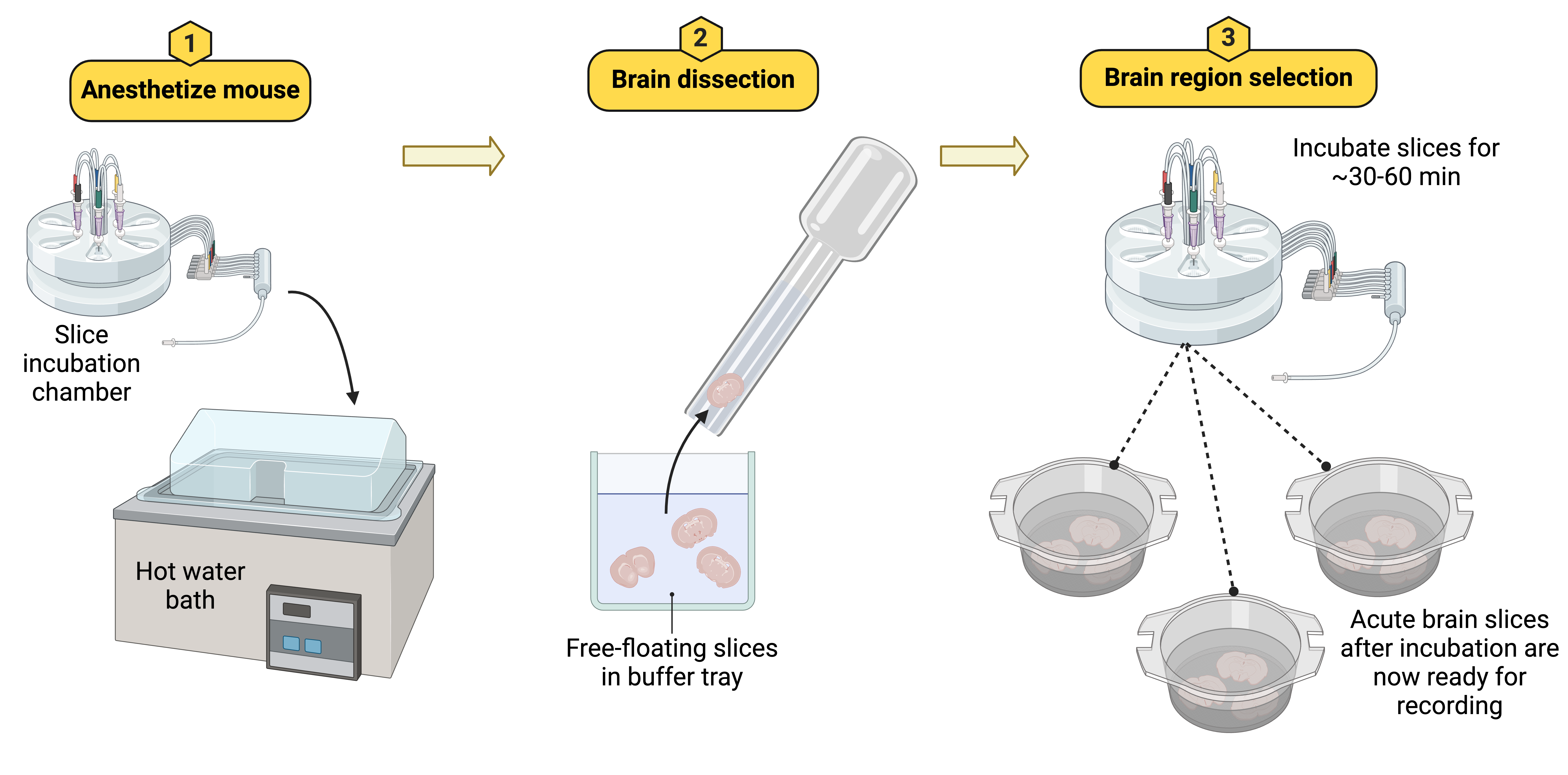
Figure 10. Acute brain slices are incubated at 37°C immediately after cutting to preserve cellular integrity and function. The slices are gently transferred into a prewarmed incubation chamber filled with ACSF, using a modified plastic pipette. After a 30-60 minute incubation period, the slices are ready for patch-clamp recording experiments.
Developed for Researchers in Neuroscience and Electrophysiology
At Precisionary Instruments, we work closely with scientists in neuroscience, physiology, and biomedical research, and one of the most frequent requests we receive is for a structured, visual guide to acute brain slicing for patch-clamp electrophysiology.
Many standard protocols lack visual aids and practical guidance on how to prepare slices effectively, leading to inconsistencies between laboratories. This guide was created to fill that gap by offering a standardized workflow that researchers can follow to improve reproducibility and efficiency in their electrophysiology experiments.
The methodology outlined in this guide is based on best practices used in laboratories worldwide, incorporating insights from scientists who specialize in patch-clamp electrophysiology. Whether you are troubleshooting persistent issues with slice quality or looking for a structured approach to introduce new trainees to the technique, this guide provides the resources you need.
Download the Full Guide for Acute Brain Slices
For researchers looking to enhance their electrophysiology experiments, this guide serves as an invaluable resource. The full protocol is now available for download, providing a detailed breakdown of every step involved in the acute brain slice preparation process.
Download the complete protocol here: https://products.precisionary.com/electrophysiologyguide/
We hope this guide helps improve the efficiency and quality of your experiments. If you have any questions, we encourage you to reach out to us. At Precisionary Instruments, we are committed to not only providing high-quality vibratome equipment but also supporting researchers with scientific expertise in tissue preparation and electrophysiology.
For further inquiries, support, or guidance on optimizing your electrophysiology workflow, contact our team at Precisionary Instruments. We are here to assist you at every stage of your research.